Irrigation with commercial bottled water increases the generation of reactive oxygen species in Nicotiana tabacum
November, 2020
Marissa Calderón-Torres, Edith López-Estrada, Ana E. Ortiz-Reyes1 & Miguel Murguía-Romero
Summary
The consumption of commercial bottled water has increased dramatically worldwide. Research is needed to ensure that its consumption does not have harmful effects on human health. The growth of Nicotiana tabacum plants was analyzed after being irrigated with tap water and commercial bottled water. Plants irrigated with commercial bottled water had fewer leaves, some with chlorosis, and shorter stem and leaf lengths compared to plants irrigated with tap water. Their roots, stems, and leaves also showed increased production of reactive oxygen species and a significant decrease in chlorophylls. Chemical analysis of commercial bottled water showed a low ion concentration and an acidic pH value (5.5), below the minimum of the Mexican standard NOM-127-SSA1-2000 (6.5–8.5). The growth, chlorophyll concentration, and antioxidant capacity of plants watered with bottled water are negatively affected compared to those watered with tap water.
Keywords: Nicotiana tabacum, commercial bottled water, chlorosis, reactive oxygen species.
Introduction
Bottled water consumption is increasing worldwide, and Mexico is one of the leading countries with the highest consumption [1]. The introduction of commercial bottled water in Mexico was historically due to water scarcity, to cover urgent needs during natural disasters, to provide water to the population in cities in desert areas, and to the presence of pathogenic microorganisms in drinking water [3,4].
The high consumption of commercial bottled water has led to the investigation of the following three essential aspects: i) the mineral and organic composition, as well as microbiological contaminants and radioactivity, because these can cause serious effects on human health [5]; in the case of bottled water in Mexico, organochlorine pesticide residues, alpha radioactivity, and the presence of pathogenic microorganisms such as coliforms have been found, all of which exceed the limits permitted for human consumption [6,7,4]; ii) the water disinfection method, since the derived components can react with organic ions dissolved in the water, such as halogen compounds or nitrosamines, which have been reported in drinking water [8,9]; finally, iii) the type of plastic in which the water is bottled, which has received special interest because the type of polycarbonate plastic and the resins used in its production [10] can cause the release of different elements and organic compounds into food or water [11] that can affect the human endocrine system [12]. For example, antimony (Sb) and phthalates have been found in commercial bottled water in the city of Monterrey, Mexico. Their presence is derived from the polyethylene terephthalate (PET) plastic used for water bottles, and both are considered toxic and harmful to the environment [13,14].
In most of these research works, the objective is to determine whether the quality of the water intended for human consumption meets the standards established by the legislation of the corresponding country to ensure that there is no risk to human health. However, direct evaluation of the effects of prolonged consumption of commercial bottled water in humans is scarce, probably because the health effects are variable, and depend on the time at which the first health problems appear, which can vary from months to years. Therefore, the use of living organisms, such as plants or animals, is preferred in the study of diseases or therapies, due to their capacity to respond or evolve to changes in their environment [15]. Plants are widely used to monitor environmental pollution and, among the plants used for this purpose, tobacco plants have proven to be useful [16]. It has recently been shown that the gene expression and specific activity of enzymes in the central metabolism of N. tabacum, for example, NADP+ malate oxidoreductase, are sensitive to various abiotic stressors, such as changes in salinity, temperature, pH, and drought, among others [17,18].
Three facts highlight the need for research into the adverse effects of bottled water consumption on humans. First, drinking water quality research that assesses health risks to Mexicans has focused only on the analysis of mineral and organic components, as well as radioactive and biological contaminants. Second, there are few studies focused on evaluating the effects of commercial bottled drinking water on living organisms. And finally, commercial bottled water consumption is high in Mexico.
To assess the potential health effects of commercial drinking water, one can begin by investigating adverse effects in animal or plant models. Plants are suitable models because they have a relatively short life cycle and have been used to investigate the effects of contaminants. This study evaluated the growth of Nicotiana tabacum plants irrigated with commercial bottled water for 55 days and compared it with that of plants irrigated with tap water, measuring morphological and biochemical parameters.
Materials and Methods
The plants were grown according to [19]. Germination and growth of Nicotiana tabacum took place in the Botanical Garden of the Iztacala School of Higher Studies, UNAM. Prior to germination, the Nicotiana tabacum seeds were scarified with NaClO (4%) and placed in a 10-mm mesh sieve, then washed with sterile distilled water. They were then placed in sterile soil (soil previously filtered through a 3-mm mesh sieve) and incubated under standard growth conditions at 25°C, 35% relative humidity, and a 12-hr light/dark cycle. After three weeks, the seedlings were transplanted into sterile soil (filtered through a 1-cm mesh sieve).
The plants were divided into two irrigation groups of five plants each: i) tap water from the Miguel Hidalgo Municipality, Mexico City, and ii) commercial bottled water for human consumption. The plants were grown for 55 days and watered every other day with 10 mL of each water type. At the end of growth, leaf length and width, as well as stem length, were measured. A sample of the root, stem, and leaf from each plant was placed in 1.5 mL Eppendorf tubes and frozen with liquid nitrogen for storage at -70 °C.

Figure 1. Nicotiana tabacum after growing in two types of water. Plants were irrigated every three days for 55 days with a) tap water (n=5) and b) commercial bottled water (n=5).
Total Cell Extracts of Nicotiana tabacum
To prepare the cell extracts, 100 mg of samples were taken from each root, stem, and leaf and placed in 1.5 mL Eppedorf tubes. 300 mL of 10 mM phosphate-buffered saline (PBS) and 3 mL of 100 mM phenylmethylsulfonyl fluoride (PMSF) were added to each tube, and the samples were homogenized with a Teflon micro-pestle at 4°C. The tubes were then centrifuged at 16,000 x g for 5 min at 4°C. The supernatant was transferred to a new Eppendorf tube and incubated at 4°C. The protein concentration in the cell extract was determined using a bovine serum albumin (BSA) protein standard curve.
Reactive oxygen species (ROS) production in Nicotiana tabacum
ROS levels in each of the three plant tissues analyzed (root, stem, and leaf) were determined using the method described in [20]. Three samples were prepared from each of the five plants for each tissue in each experimental group, i.e., 90 readings in total. This method is based on the reaction of hydrogen peroxide with the compound 2'7'-dichlorodihydrofluorescein diacetate (DCFH-DA), which produces 2'7'-dichlorofluorescein with fluorescence emission. The reactions were performed in a 96-well plate; 20 mL of cell extract, 225 mL of 10 mM PBS, and 5 mL of DCFH-DA (500 mM) were added to each well. Emission readings were recorded every 20 min for 1 h on a fluorescence spectrophotometer (Bio Tek) at 485 nm excitation and 520 nm emission. The maximum fluorescence value obtained in each time series was used as an index of ROS production per mg of total protein.
Chlorophyll Quantification
Chlorophylls were measured using the method of [21]. First, 100 mg of leaf from each plant was taken and placed in a 1.5 mL Eppendorf tube, then 100 mL of acetone (80%) was added, and the mixture was macerated with a Teflon micropestle. The macerate was transferred to a 15 mL Falcon tube and diluted with 2 mL of acetone (80%). The tube was vortexed and centrifuged at 500 x g for 10 min. One mL of chlorophyll extract was added to a quartz cell, and the absorbance reading was recorded on a spectrophotometer at 645 and 663 nm. The absorbance data obtained at each wavelength were used to calculate the chlorophyll concentration in mg/mL.
Antioxidant Capacity of Nicotiana tabacum
The total antioxidant capacity of Nicotiana tabacum leaves was determined using a commercial system (Cayman Chemical, No. 709,001). The system relies on an oxidation reaction in which the enzyme metmyoglobin and hydrogen peroxide (H2O2) produce the oxidation of 2'2-azino-bis-[3-ethylbenzothiazole-6-sulfonic acid] (ABTS) with a color change; however, the antioxidants present in the cell extracts prevent the oxidation of ABTS. In this study, the enzyme peroxidase (Pepro Tech) was used instead of the metmyoglobin enzyme. The antioxidant capacity of the cell extracts was determined using a TROLOX standard curve. This compound is an analogue of tocopherol or vitamin E. Oxidation reactions were performed in a 96-well plate. 10 mL of cell extract (volume adjusted to a total protein concentration of 1.5 mg/mL), 10 mL of the peroxidase enzyme, 40 mL of hydrogen peroxide, and ABTS were added to each well. The absorbance of the plate was read using a spectrophotometer at 405 nm. Antioxidant capacity was expressed in equivalents of the molar concentration of TROLOX.
Water Sampling
Bottled drinking water of a commercial brand was purchased from retail stores (n = 3) in the Miguel Hidalgo Municipality of Mexico City, and tap water was collected from a residential intake in the same area (n = 3). At the time of purchase of the water bottles, it was verified that the expiration date was at least six months in the future. The protocols for the collection, preservation, and transportation of tap and bottled water samples followed the procedures described in [22]. After collection, all samples were immediately transported to the Analytical Chemistry Laboratory of the Institute of Geophysics, UNAM (a laboratory specialized in water analysis) and kept refrigerated until analysis.
Chemical Analysis of Water
The chemical composition of the tap and bottled water was determined in triplicate in the Analytical Chemistry Laboratory of the Institute of Geophysics, UNAM. The analysis included pH, conductivity (ms/cm), sodium, potassium, calcium, and magnesium cations, as well as carbonate, sulfate, chloride, boron, and nitrate ions (mg/L). This specialized laboratory carried out all chemical analyses according to standard procedures established by the American Public Health Association.
Quantification of Na+ and K+ in Nicotiana tabacum
Na+ and K+ were determined as described in a previous study [19]. A 200 mg sample of root, stem, and leaf tissue from the five plants in each irrigation group was dehydrated in a drying oven at 80°C for 48 hours. Each cold sample was then weighed and placed in a 15 mL glass test tube, to which 10 mL of Milli Q water was added. The tubes were incubated in a boiling water bath for 1 hour. After cooling to room temperature, the tubes were centrifuged at 1800 x g for 10 minutes. The supernatant was transferred to a 15 mL Falcon tube. Each ion was determined with 1 mL of the supernatant in a flame photometer (Zeiss PF5). A solution of 1 mM NaCl and 1 mM KCl was used as a standard for each ion. From the flame photometer readings, the sodium and potassium concentrations were calculated and expressed as mol/L.
Statistical Analysis
The mean value and standard error of the mean (SEM) were obtained for each determination. The statistical significance of the differences between the two irrigation groups was assessed using the Student t test.
Results
Morphological parameters of N. tabacum irrigated with tap water and bottled water
The average leaf size of the plants was significantly different (Table 1). The leaf length of plants irrigated with commercial bottled water was shorter compared to that of plants irrigated with tap water (length 7.1 cm vs. 10.5 cm), and stem length was also significantly different (7.8 vs. 12.3 cm). The average number of leaves was higher in plants irrigated with tap water (10.6 vs. 8.0). In addition, chlorosis was observed on the leaves of plants grown with commercial bottled water (Figure 1).
ROS Production and Antioxidant Capacity in N. tabacum Cell Extracts
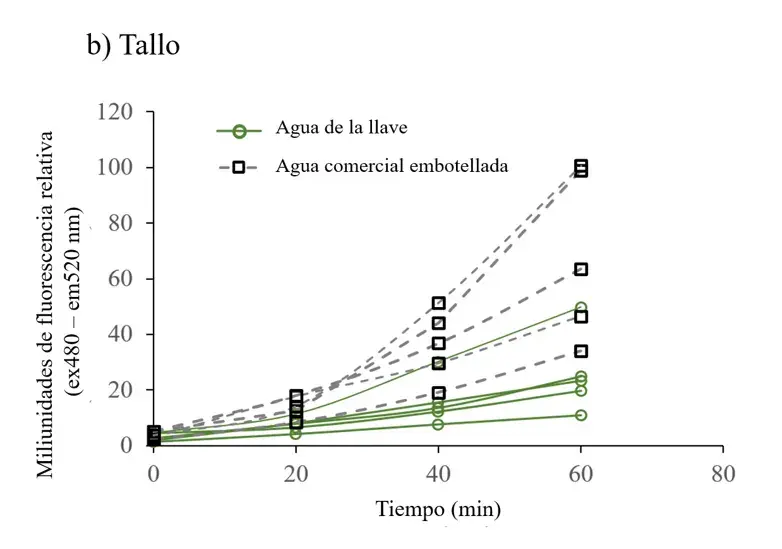
To determine whether the chlorosis observed in leaves of plants grown with commercial bottled water correlated with free radical production, the concentration of reactive oxygen species was measured in all plants. In plants irrigated with commercial bottled water, ROS levels increased over time in all plant structures (Figure 2), with particular significance in the stem (Figure 3a).
Regarding antioxidant capacity, leaves of plants irrigated with commercial bottled water exhibited reduced antioxidant systems compared to plants irrigated with tap water (Figure 3b).
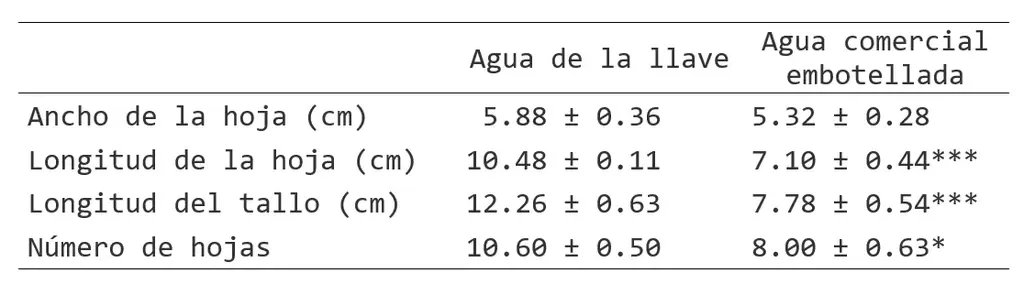
Table 1. Morphological measurements of stem and leaf of plants irrigated with tap water and commercial bottled water. Mean values ± SEM; n=3. *P<0.05, ***P<0.001.
Figure 2. ROS fluorescence time series of cell extracts from root (a), stem (b) and leaf (c) of plants irrigated with tap water and commercial bottled water.
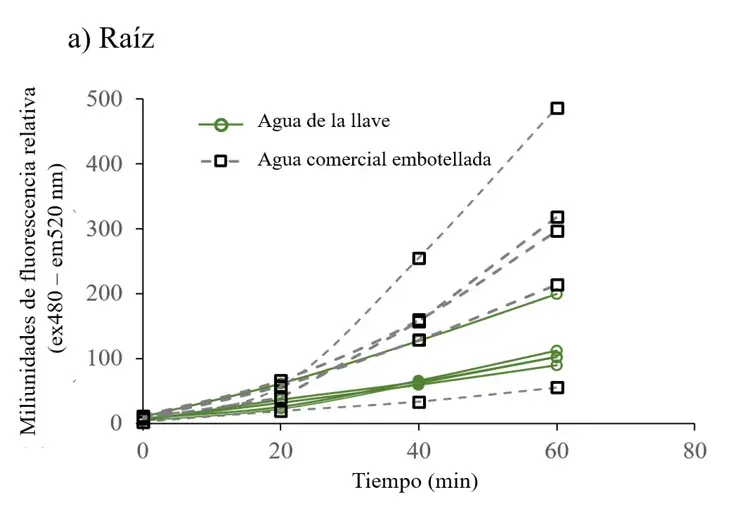
Cell extracts were read with 10 M DHFC-DA, and fluorescence (excitation at 485 nm and emission at 520 nm) was recorded every 20 min for 60 min. Differences in fluorescence were most evident at 60 min: a) ROS fluorescence data in cell extracts from root, b) stem, and c) leaf. Each fluorescence data point is the average of three readings and was adjusted to the total protein concentration. Each line represents data from one plant.

Soluble Na+ and K+ Ions in Nicotiana tabacum
To explore possible causes of chlorosis, the concentration of sodium and potassium ions in the roots, stems, and leaves was quantified. Na+ concentrations showed a significant increase in the roots and leaves of plants irrigated with commercial bottled water (Figure 4a), while there was a higher sodium concentration in the stems of plants irrigated with tap water.
Plants irrigated with commercial bottled water had higher potassium concentrations (Figure 4b). This difference was statistically significant in the roots and leaves.
Chlorophyll A, B, and Total Quantification
To corroborate whether the leaves of the plants showed a change in the proportion of chlorophyll, their concentration was measured. The leaves of plants irrigated with commercial bottled water had lower concentrations of chlorophylls A, B, and total compared to plants irrigated with tap water. The differences were significant for chlorophyll A and total (Figure 3c).
Figure 3. ROS, antioxidant capacity, and chlorophylls in Nicotiana tabacum.
a) Reactive oxygen species (ROS) in roots, stems, and leaves of plants irrigated with tap water and commercial bottled water; b) antioxidant capacity in leaves; c) chlorophylls A, B, and total in leaves (n=5 for each group). Error bars represent the standard error of the mean. *P < 0.05.
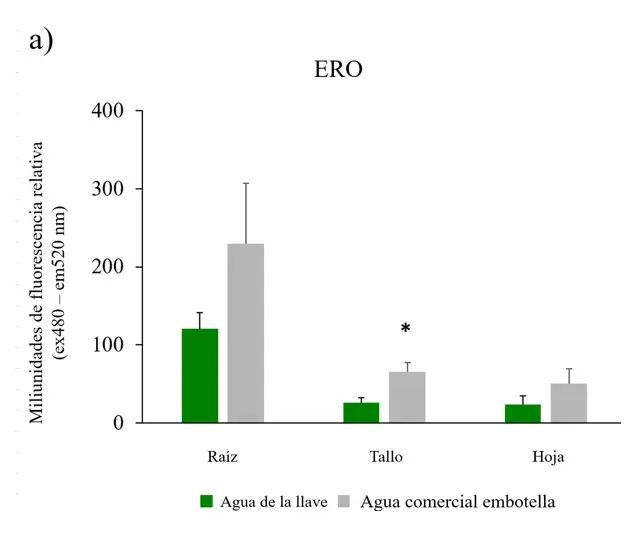
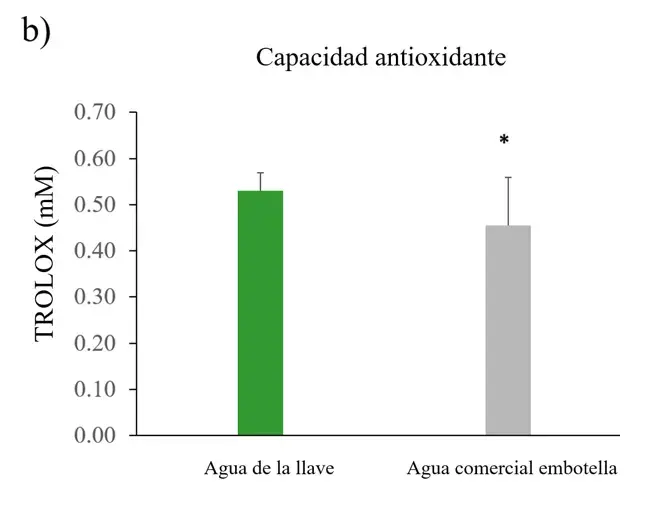
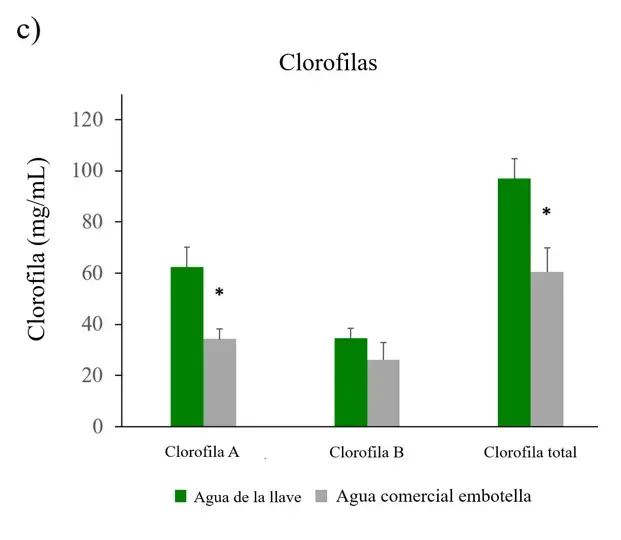
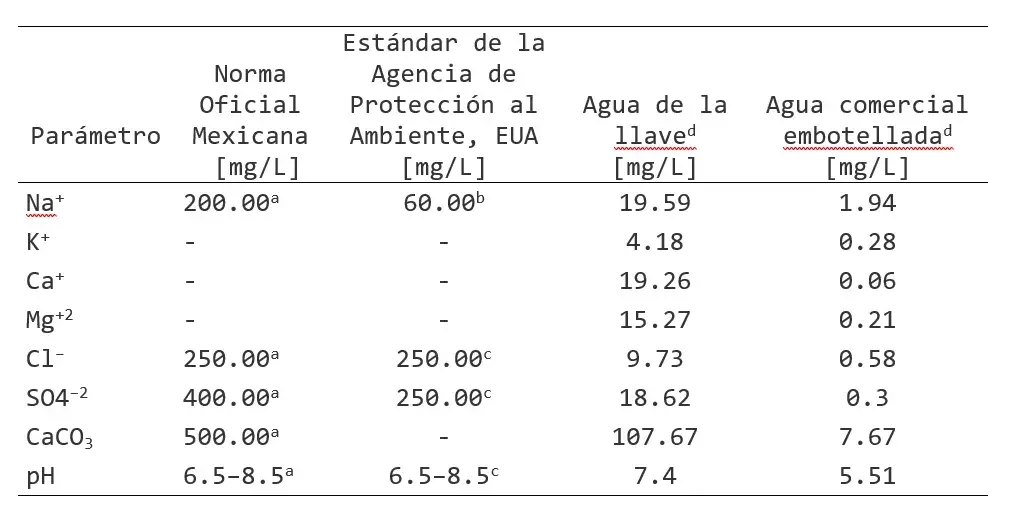
Table 2. Basic chemical composition of the water used to irrigate Nicotiana tabacum.
a) Maximum allowable limit according to the NOM-127-SSA1-2000 standard in Mexico;
b) Recommended limit concentration for flavor;
c) Maximum allowable limit according to the secondary water quality regulation of the U.S. Environmental Protection Agency (EPA) 822-F-18-001 2018 Drinking water standards and health advisories tables;
d) Average concentration; n=3.
Chemical Analysis of Water
The basic ion composition of both types of water was determined. Table 2 shows that the ion concentration in commercial bottled water was lower compared to that of tap water. Furthermore, an acidic pH value (5.5) was observed in commercial bottled water.
Discussion
In this study, changes in the growth of Nicotiana tabacum were observed after irrigation with tap water and commercial bottled water. Plants irrigated with commercial bottled water were significantly smaller and exhibited yellowing of the leaves, known as chlorosis. This condition is caused by a lack of chlorophyll. Chlorophyll concentration in the leaves was measured to corroborate the low levels. As expected, plants irrigated with commercial bottled water contained less chlorophyll A, B, and total chlorophyll. Chlorophyll content at different stages of the tobacco leaf is one of the main biomarkers of senescence [25]. Therefore, the presence of chlorosis in young leaves of N. tabacum irrigated with commercial bottled water indicates a serious problem in leaf physiology that leads to senescence.
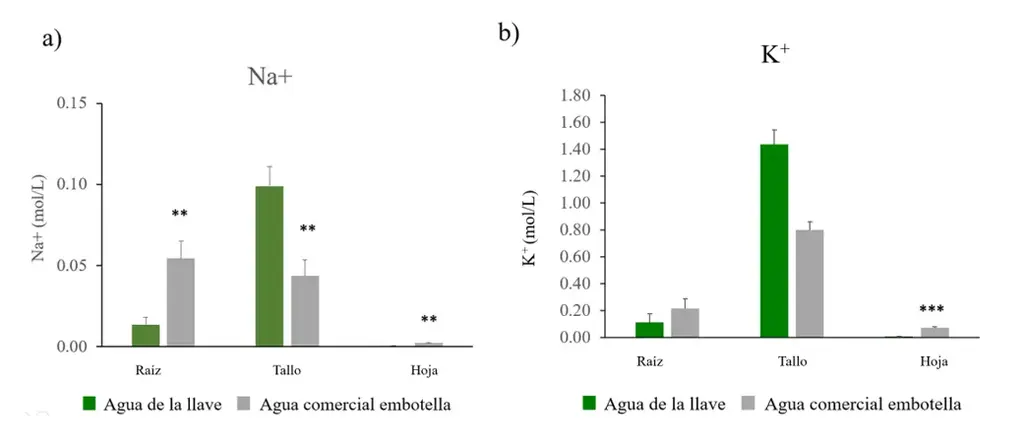
Figure 4. Na+ and K+ concentrations in Nicotiana tabacum after irrigation with tap water and commercial bottled water. Quantification of a) sodium and b) potassium in the roots, stems, and leaves of plants from each group: tap water (n=5) and commercial bottled water (n=5). Bars represent the standard error of the mean. **P < 0.01, ***P <0.001.
It has been shown that under various stress conditions such as drought, flooding, frost, increased salinity, etc., chlorophyll degrades, leading to leaf chlorosis [26] with subsequent leaf loss. However, this fact is only the tip of the iceberg; chlorosis is a consequence of stress.
Therefore, the next step in this work was to investigate the cellular stress that leads to chlorophyll degradation and, consequently, chlorosis. To this end, it was considered that under different stress conditions, a common factor is the overproduction of reactive oxygen species (ROS) and nitrogen species (ENO) [27, 26] with the loss of antioxidant capacity, resulting in damage to biomolecules [29, 30]. Therefore, in this study, ROS production was measured by its reaction with the compound 2'7'-dichlorodihydrofluorescein diacetate (DCFH-DA), commonly used to quantify H2O2 [31, 32], and all three types of plant tissues analyzed irrigated with commercial bottled water had elevated ROS levels. Antioxidant capacity was also quantified in leaf extracts from these plants, and a decrease was observed.
Antioxidant capacity is generally assessed by the reaction of the commercial enzyme metmyoglobin and hydrogen peroxide (H2O2), which oxidize ABTS, but this reaction is prevented by the antioxidants present in cell extracts. However, in this study, the reaction was carried out with the enzyme peroxidase and H2O2; ABTS oxidation also occurs, and the ABTS+ radical is formed. However, if catalase enzymes are present in the cell extracts, the peroxide is transformed into water and oxygen, which decreases the formation of ABTS+ [33]; Therefore, a low absorbance value corresponds to a higher concentration of TROLOX, i.e., a high antioxidant capacity. Measuring antioxidant capacity with the enzyme peroxidase also indicates peroxide formation, and the highest concentration of TROLOX was observed in cell extracts from the leaves of plants irrigated with tap water, indicating that antioxidant enzymes are degrading H2O2. In contrast, cell extracts from the leaves of plants irrigated with commercial bottled water showed lower concentrations of TROLOX and elevated levels of ROS. This likely corresponds to damage to antioxidant enzymes, which are responsible for the degradation of superoxide anion and hydrogen peroxide.
To verify the presence of hydrogen peroxide and catalases in cell extracts, we propose that further studies measure hydrogen peroxide by radical scavenging activity in the presence of a scavenging agent, e.g., caffeic acid [34], which acts as a hydrogen peroxide antagonist by donating hydrogen atoms to reduce hydrogen peroxide to water. And to determine the presence of the catalase enzyme in plants, we propose measuring the antioxidant capacity in the presence and absence of a catalase enzyme inhibitor such as the compound 3-amino-1,2,4 triazole, which has been used in adipocytes to unbalance the antioxidant defense system by increasing the H2O2 concentration [35].
Plants normally have efficient mechanisms to neutralize ROS [36]. If the concentration of antioxidant molecules and enzymes decreases and reactive oxygen species increase, an imbalance occurs between ROS production and antioxidant capacity. Overproduction of ROS is potentially harmful to proteins and lipids in chloroplasts. Therefore, irrigation with commercial bottled water is considered to lead to severe oxidative stress with an increase in ROS, which is associated with the chlorosis observed in the leaves.
It is clear that low potassium levels in commercial bottled water may be a key factor in leaf chlorosis, as well as the low potassium concentration observed in the stems of these plants. In the case of magnesium, this cation is a key component of chlorophyll molecules involved in light absorption. Some studies have reported that Mg+2 deficiency triggers leaf chlorosis; A mechanism has recently been proposed in which ROS are produced via photooxidation due to an accumulation of carbohydrates and failure of CO2 fixation, which in turn can accelerate the transfer of photosynthetic electrons to O2 and enhance ROS production [40,41]. Secondly, the acidic pH of water can change the characteristics of the soil and consequently alter various plant functions. At the beginning of irrigation with commercial bottled water, it is likely that the soil composition buffers the acidic pH of the water as well as within the cell, since it has been shown that the pH of the cytoplasm of plants under normal conditions is 7.5, i.e., slightly alkaline [42,43]. However, after prolonged irrigation with water with acidic pH, it is very likely that the soil pH balance is altered, affecting plant physiology. It has been proven that short-term exposure of soils or growth media to acidic pH can modify root permeability; for example, the activity of specialized water transport proteins aquaporins in the root can be reduced, consequently modifying the transport of nutrients and water from the root to the leaf [44,45]. Thus, prolonged exposure to acidic pH can reduce plant growth, and this may be the case for plants irrigated with commercial bottled water. Both mineral deficiency and acidic water pH can be the cause of increased ROS production, reduced chlorophyll levels, and consequently, small plant sizes.
We propose that subsequent studies examine the concentration of oxidative stress markers such as malondialdehyde and 3-nistrityrosine at different plant growth stages. To examine the accumulation of hydrocarbon-derived compounds, ROS formation can be assessed using the dithiothreitol assay [46]. Furthermore, the expression of the NADP+ malate oxidoreductase enzyme can be quantified as a sensor of changes in soil pH.
The commercial bottled water analyzed in this study did not comply with Mexican regulation NOM-127-SSA1-2000, which establishes a standard pH value of 6.5–8.5. It is considered that if the adverse effects observed on the physiology of plants irrigated with commercial bottled water are extrapolated to continuous human consumption, this could have serious health implications. The first clear effect may be the alteration of acid-base homeostasis that can lead to a low-grade metabolic acidosis, characterized by a decrease in blood pH (<7.35). This has been associated with the type of diet, for example, meats and cheese are foods that induce the production and constant increase of acid, which in turn is associated with an increased risk of developing cardiovascular disease, insulin resistance, high blood pressure and changes in bone mineral density [47,48]. Since the production of ROS is a common factor in all organisms with aerobic metabolism, we consider that the overproduction of ROS observed in plants irrigated with commercial bottled water is an important element that should be extrapolated to the effects on human health. Under normal conditions, ROS have a beneficial effect, functioning as a secondary messenger in various cellular processes, for example, in plants they have an effect on stomatal closure [49,50]. On the contrary, excess ROS induce oxidative damage under severe environmental stress conditions. In humans, ROS overproduction is a key element in the theory of aging, and is a marker of pathology and progression of different chronic degenerative diseases such as diabetes, chronic obstructive pulmonary disease, osteoarthritis, and some cancers [51–54]. Therefore, it can be hypothesized that prolonged intake of acidic water results in increased ROS production with harmful effects on human health.
Conclusions
In this study, we found that irrigating Nicotiana tabacum with commercial bottled water leads to reduced plant size and leaf chlorosis. The study of the plants showed a significant increase in reactive oxygen species and a decrease in chlorophyll A and total chlorophyll, and their leaves had a lower antioxidant capacity. Together, these factors may cause the physiological alterations observed in plants irrigated with commercial bottled water. If these effects are extrapolated to human health, such as the loss of acid-base balance and overproduction of ROS, drinking bottled water may accelerate the development, pathology, and progression of some chronic diseases.
References
Highlighted Results